Case Info
Philips Recalls Millions of Breathing Machines
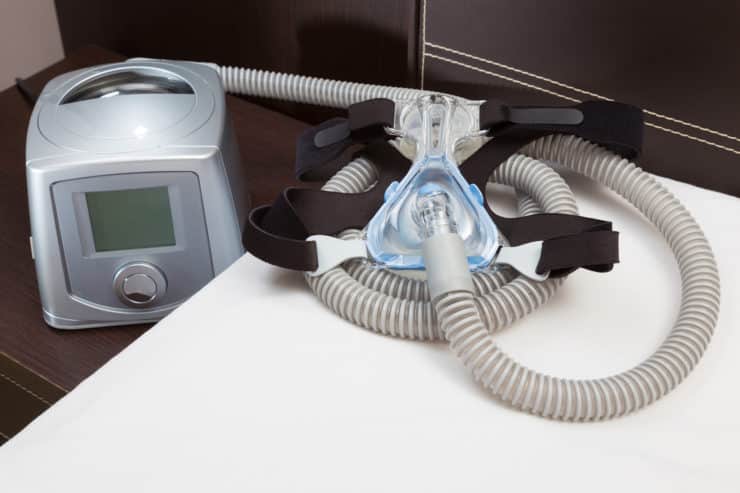
On June 14, 2021, Philips announced a recall of more than 10.8 million CPAP, BiPAP, and mechanical ventilator devices because of the potential health risks of defective foam in the devices. The polyester-based polyurethane (PE-PUR) foam used in these devices was found to degrade into small black particles and release toxic chemicals into the air pathways. Inhalation or ingestion of these particles and chemicals could lead to injuries such as serious respiratory problems and cancer.
The U.S. Food and Drug Administration (FDA) classified the recall as a Class I recall, the most serious type, indicating a high probability of serious injury or death. Since the recall, the FDA has received over 116,000 complaints related to the recalled Philips CPAP devices, including at least 561 reports deaths.
Plaintiffs File Lawsuits Against Philips
Following the recall, patients across the United States filed individual personal injury lawsuits seeking compensation for injuries caused by defective Philips devices. Additionally, several class-action lawsuits were initiated to recover economic damages for the cost of replacing these devices and to secure medical monitoring for potential future health issues. These legal actions were consolidated by the Judicial Panel on Multidistrict Litigation in the Western District of Pennsylvania before the Senior U.S. District Court Judge Joy Flowers Conti on October 8, 2021.
Seeger Weiss was among the first law firms to file lawsuits against Philips regarding the defective foam and represented thousands of people seeking justice. Founding partner Chris Seeger was appointed as co-lead counsel by Judge Conti in the consolidated litigation, In Re: Philips Recalled CPAP, Bi-Level PAP, and Mechanical Ventilator Products Litigation.
According to internal documents uncovered during the litigation, Philips may have known about the potential risks of the devices as far back as 2015. In April 2018, three years before the first recall, a Philips engineering employee contacted the supplier of the foam used in the devices. Emails and customer reports show concerns over foam degradation and the potential for inhalation of particles.
Philips CPAP Lawsuit Results
On September 7, 2023, Seeger Weiss secured an uncapped settlement worth a minimum of $479 to resolve economic loss claims of users and payers impacted by the recall. About 3 million class members have registered for benefits, and 1.6 million have returned their devices for payment.
After receiving final approval for the economic loss settlement, the Seeger Weiss team negotiated additional agreements in April 2024 to resolve plaintiffs’ personal injury claims for $1.075 billion and medical monitoring claims for $25 million, providing justice and compensation to patients who suffered significant physical injuries caused by the recalled machines.